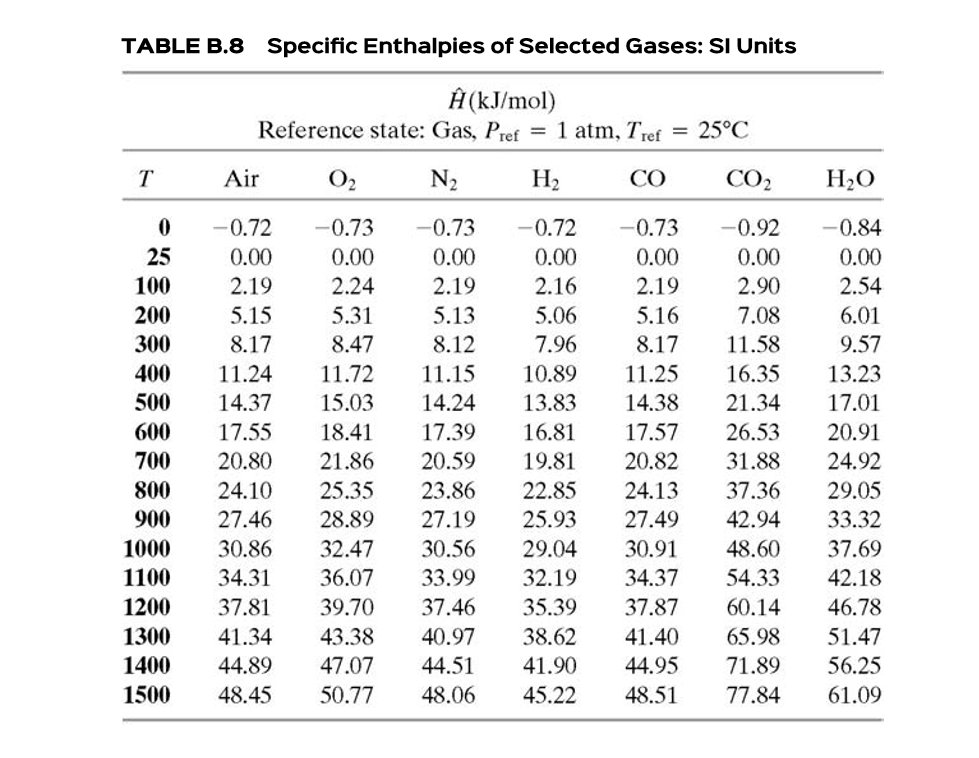
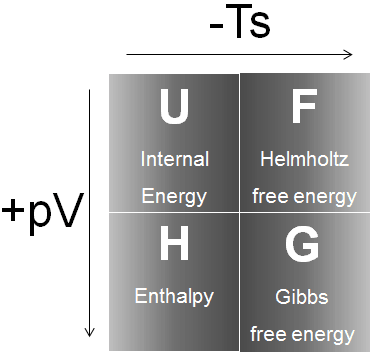
3 Enthalpy. Units SI unit = joule 1KJ = 1000J = cal 1st law of Thermodynamics The total energy of the universe is constant i.e energy cannot be. - ppt download

5 Thermochemistry 1 Copyright (c) The McGraw-Hill Companies, Inc. Permission required for reproduction or display. - ppt download