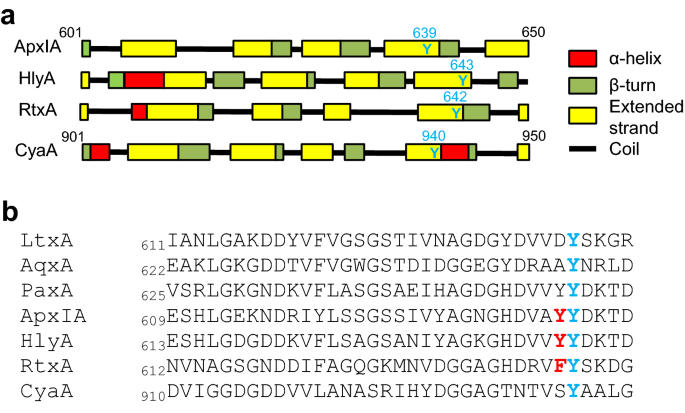
Different roles of conserved tyrosine residues of the acylated domains in folding and activity of RTX toxins | Scientific Reports

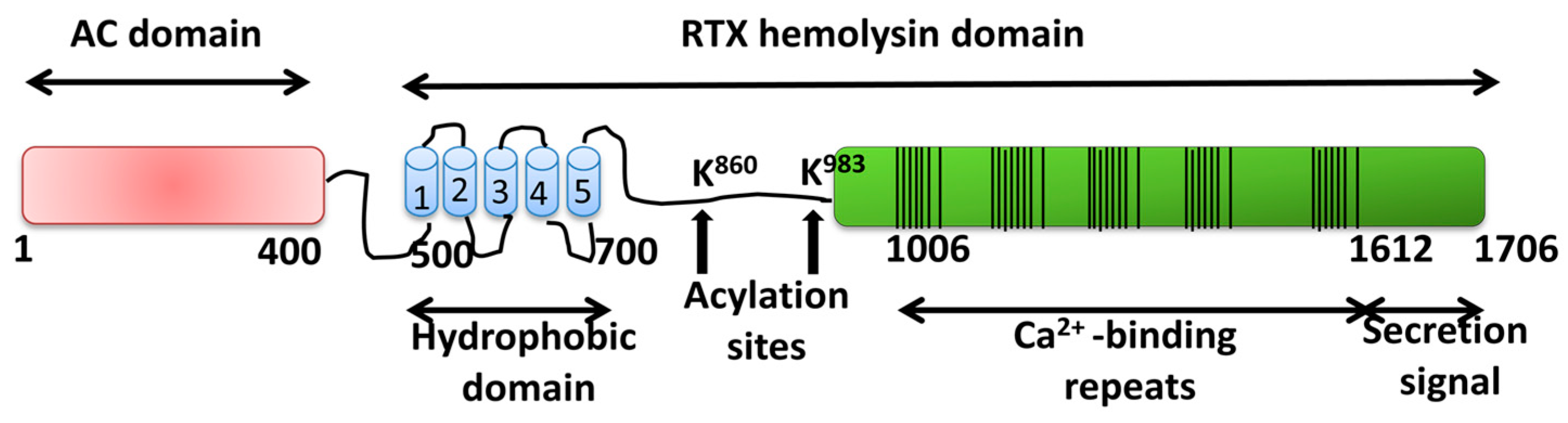
2 Schematic organization of HlyA and CyaA RTX toxins. The different... | Download Scientific Diagram

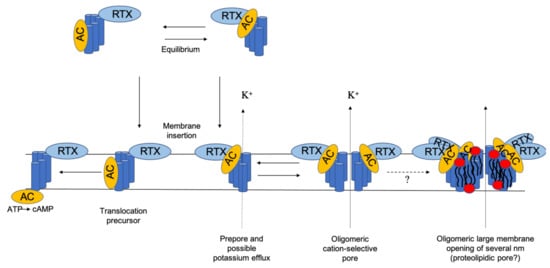
4 Schematic model for the secretion pathway of RTX toxins. Proposed... | Download Scientific Diagram

The Actin Cross-linking Domain of the Vibrio cholerae RTX Toxin Directly Catalyzes the Covalent Cross-linking of Actin* - Journal of Biological Chemistry

Channel formation by RTX-toxins of pathogenic bacteria: Basis of their biological activity - ScienceDirect

Single Molecule Force Spectroscopy Reveals the Mechanical Design Governing the Efficient Translocation of the Bacterial Toxin Protein RTX | Journal of the American Chemical Society

Bacterial RTX Toxins Allow Acute ATP Release from Human Erythrocytes Directly through the Toxin Pore* - Journal of Biological Chemistry
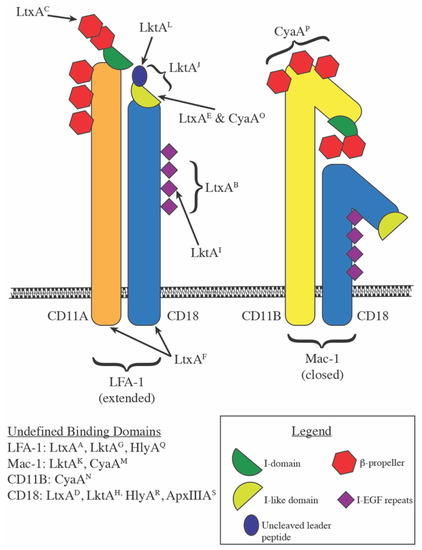
Structural basis for antibody binding to adenylate cyclase toxin reveals RTX linkers as neutralization-sensitive epitopes | PLOS Pathogens

The Vibrio cholerae MARTX toxin simultaneously induces actin collapse while silencing the inflammatory response to cytoskeletal damage | bioRxiv

The RTX pore-forming toxin α-hemolysin of uropathogenic Escherichia coli: progress and perspectives. | Semantic Scholar

Pore forming activity of the potent RTX-toxin produced by pediatric pathogen Kingella kingae: Characterization and comparison to other RTX-family members - ScienceDirect

Templated folding of the RTX domain of the bacterial toxin adenylate cyclase revealed by single molecule force spectroscopy | Nature Communications

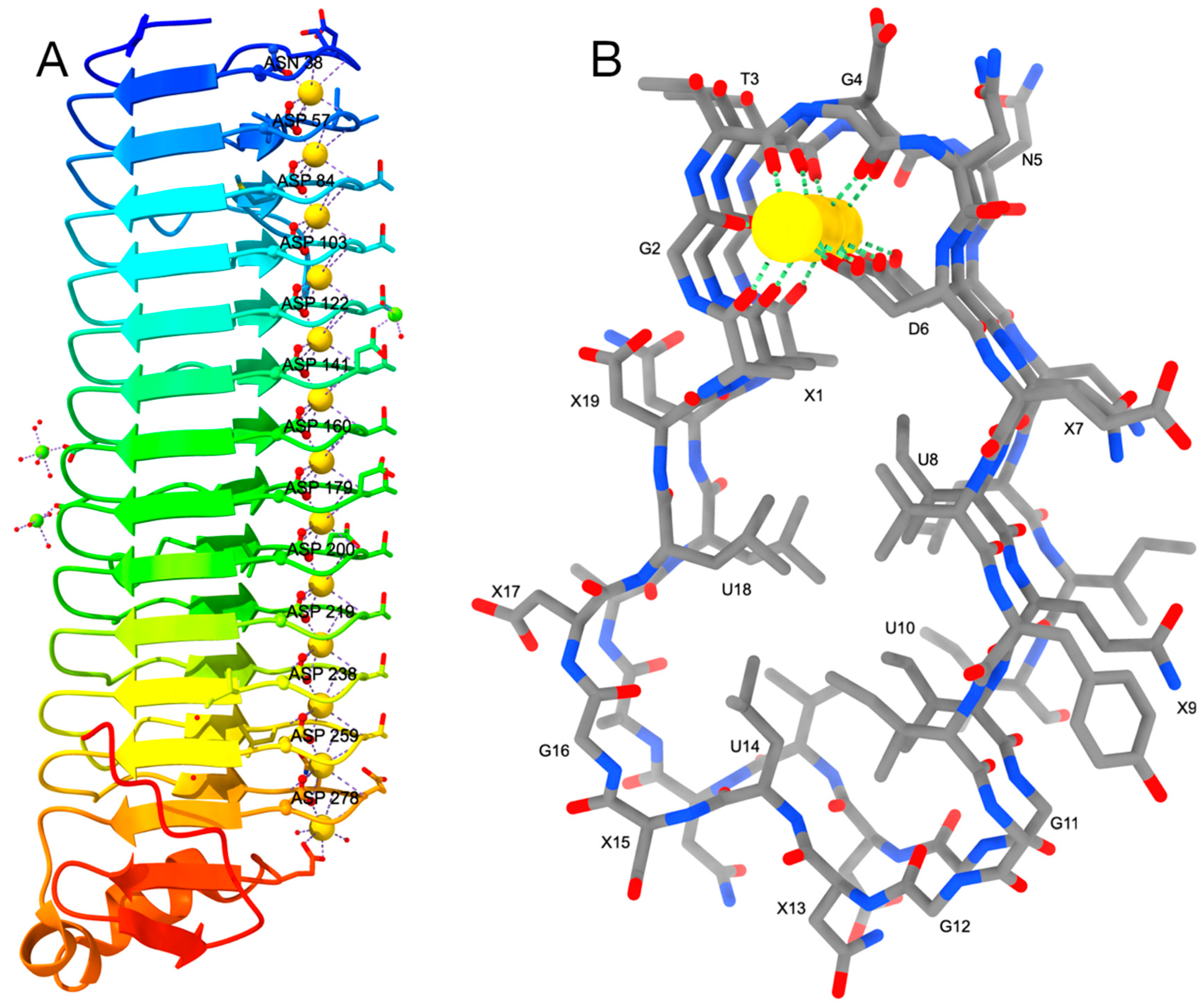
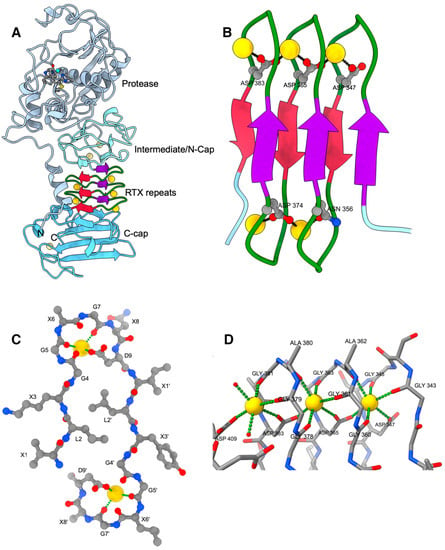
Toxins MDPI on Twitter: "Structure–Function Relationships of the Repeat Domains of #RTX Toxins https://t.co/JZ5sS9O77e #calcium; #proteinfolding; #tertiarystructure #mdpitoxins https://t.co/49AAZ478C5" / Twitter

Channel formation by RTX-toxins of pathogenic bacteria: Basis of their biological activity - ScienceDirect
Toxins | Free Full-Text | Membrane Permeabilization by Pore-Forming RTX Toxins: What Kind of Lesions Do These Toxins Form?